- Quality
- Compliance
- Integration
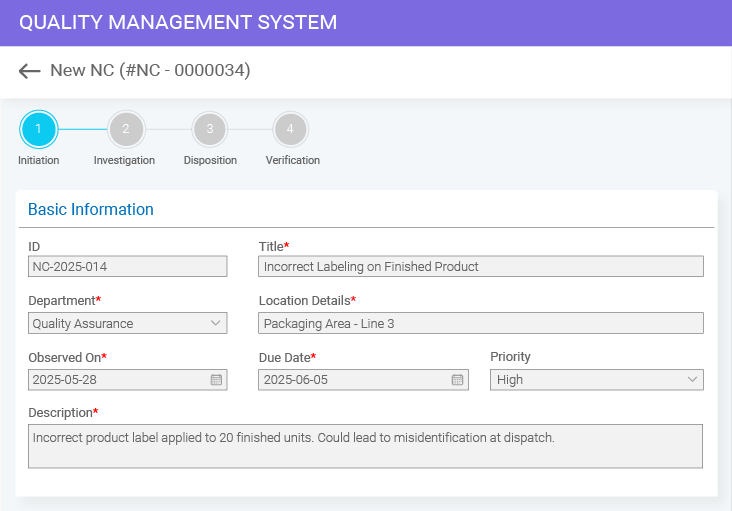
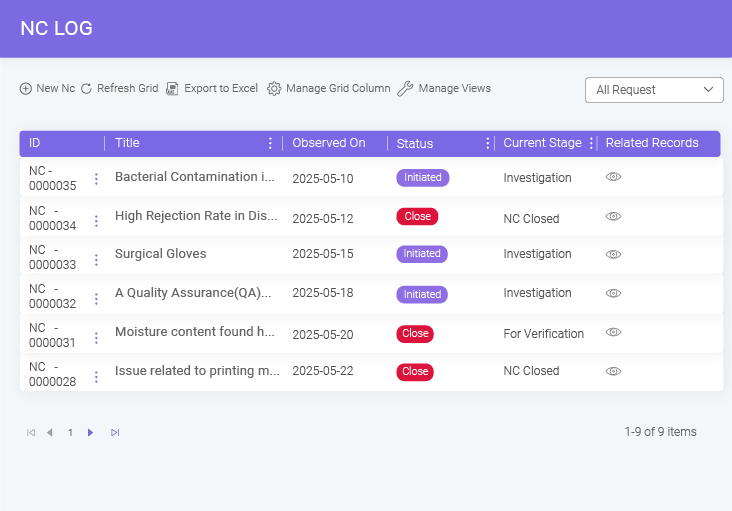
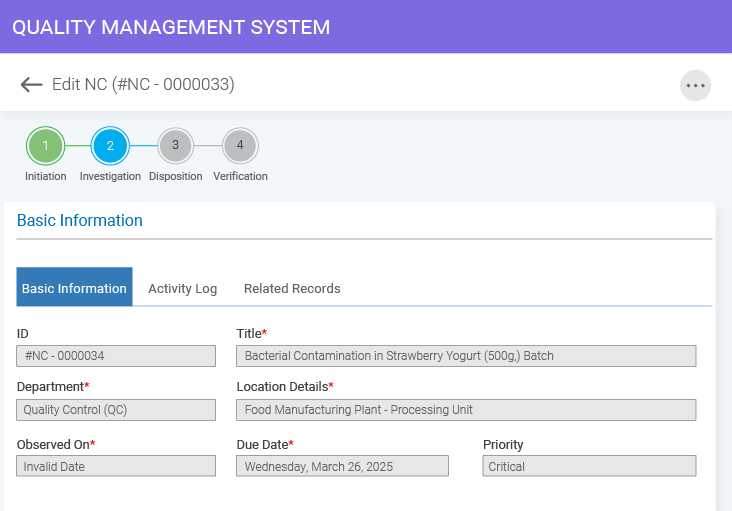
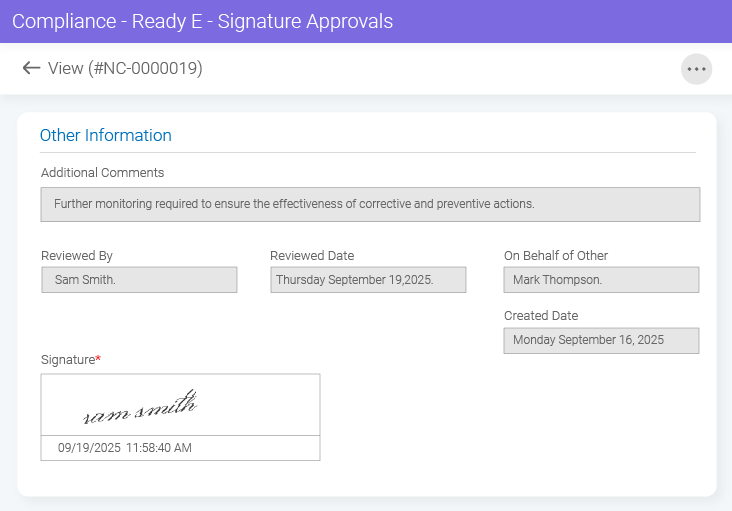
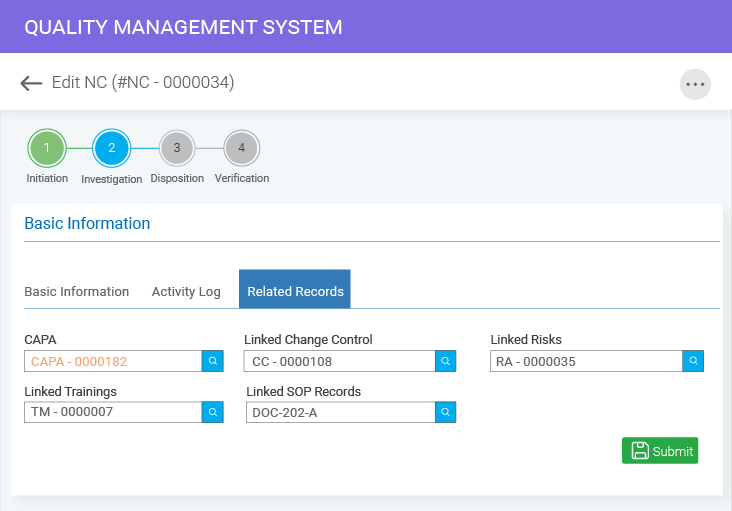
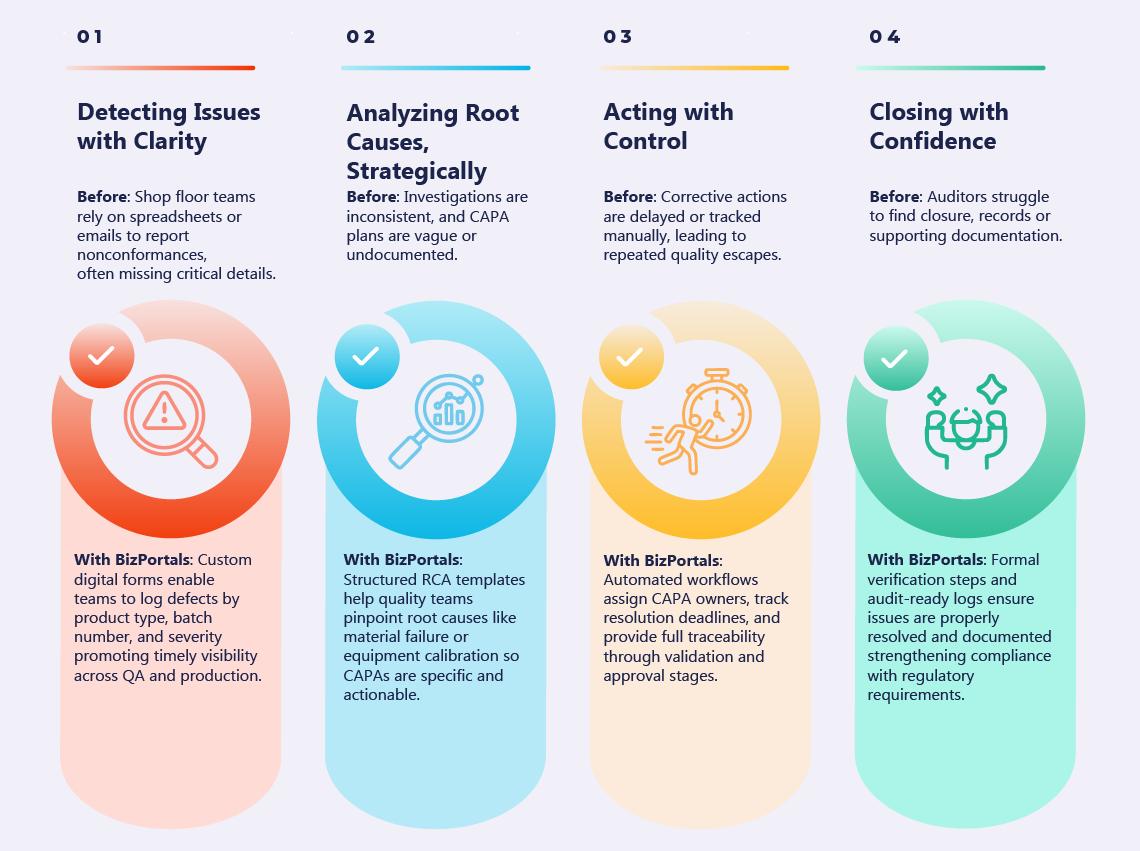
- BizPortals QCFlow (QMS) for Acumatica Unifying quality and operations for better results
- Services
- Pricing
- Resources
- Blog A treasure trove: Bridging digital expertise
- Brochures Check out our interesting brochure archive
- Guides Resourceful guides: Explore, learn, and thrive
- Whitepapers In-depth insights, research, and industry analysis
- Datasheets Core technical specs and product details
- Webinars Expert-led sessions on modern quality management
- About
- About Us Empowering businesses with transformative solutions
- Partner Program Partnering for mutual success