Summary:
This blog explores five overlooked challenges that weaken CAPA in the pharmaceutical industry, including poor root cause analysis, inconsistent documentation, cross-functional gaps, reactive practices, and a lack of digital integration. It explains why these issues lead to recurring deviations and compliance risks and outlines practical steps to strengthen investigations, collaboration, and overall quality systems.
Corrective and Preventive Actions (CAPA) in the pharmaceutical industry is a structured framework for identifying and resolving the root cause of a quality issue and preventing similar issues again.
It is a combination of systematic corrective and preventive steps that offer both immediate fixes as well as preventive measures.
Why is CAPA important in the pharmaceutical industry?
In pharmaceuticals, every quality event, no matter how small, can lead to patient safety, regulatory compliance, product availability, and even business continuity.
Primarily, drugs impact patient safety and must be consistent, pure, and potent. However, quality events can arise in the form of drug contamination, potency failures, stability failures, or mislabeling that need a strong CAPA process to prevent their recurrence.
Over the past decades, several high-profile drug shortages have been directly linked to manufacturing and product quality issues. Such issues are related to weak quality systems, inadequate root cause analysis, and most importantly, ineffective CAPA.
And this isn’t guesswork. According to FDA reports, in 2020 itself, 62 percent of all drug shortages were caused by manufacturing and product quality problems
Here is what you should focus on:
- If your quality system is solid, why are the same deviations still coming back?
- Why are major issues showing up only during audits, instead of being caught by your CAPA process earlier?
- And if everything is documented, why is your evidence still scattered across emails, shared folders, or personal notepads?
Let’s talk about a few core challenges that pharmaceutical companies face and how to resolve them.

Strengthen Your Quality Management Foundation
See how structured systems improve CAPA
View QMS FeaturesWhy Pharma CAPA Systems Fail: 5 Key Challenges and Practical Solutions
1. Superficial or incorrect root cause analysis
The primary objective of CAPA is to determine the true source of the problem, and effective RCA helps achieve this goal.
Superficial or incorrect RCA does not fully explain what happened, and the same issue resurfaces after a month or two. Sometimes in a different batch, sometimes in a different line, but with the same underlying failure.
It is not that your teams are not performing root cause analysis, but they often stop the investigation as soon as they find a convenient explanation.
If it is true, your investigation will always remain focused on immediate failure rather than tracking the issue through the entire manufacturing and quality management workflow.
You can cite many reasons for this.
An investigator may lack deep process understanding, for example, not recognizing how a minor equipment drift, an undocumented operator workaround, or a subtle raw-material variability could trigger the event.
Also, there is a silent factor that works unintentionally behind such negligence. Maybe a fear of broader implications, such as a full-scale impact assessment, employee retraining, or the requalification of suppliers or equipment.
In each case, the RCA might look complete, but it fails to map the full sequence of events, leaving the organization exposed to repeat deviations.
Here’s how to fix it:
- Train your team on process science, equipment behavior, and raw material variability, and help them understand end-to-end manufacturing and quality workflows so they can come to meaningful conclusions.
- Use structured RCA methodologies such as 5-Why, Fishbone, or the Human Error Root Cause framework, and create custom investigation templates to ensure they follow a systematic process.
- Require cross-functional reviews with Production, QC, Engineering, and Supply Chain to trace issues across the full quality workflow and not just the visible point of deviation.
- Centralize evidence, trends, and historical deviations in a digital QMS to enable investigators to identify recurring patterns and make informed decisions.
- Encourage a culture that supports identifying systemic issues instead of avoiding them.
2. Poor documentation and evidence management
With incomplete documentation and weak evidence, even a technically sound CAPA becomes unreliable, as the facts behind it feel incomplete and scattered.
Every CAPA depends on what is documented, how it is captured, and how quickly the evidence can be retrieved. This helps investigators visualize the complete picture and track the actual part of the process that is truly affected.
In the pharmaceutical industry, this becomes even more critical. Evidence such as deviation records, chromatogram reports, sample-movement logs, LIMS (Laboratory Information Management System) raw data, and OOT (Out of Trend) results must all be properly documented and fully traceable to support an effective CAPA.
Furthermore, inconsistency in documentation is also a serious issue that teams often face. Sometimes the same problem is recorded by different teams or departments in different formats.
For example, production might report “mixing anomaly” while the QC team records “viscosity outlier,” yet they both refer to the same quality event.
You can follow these steps to make it right:
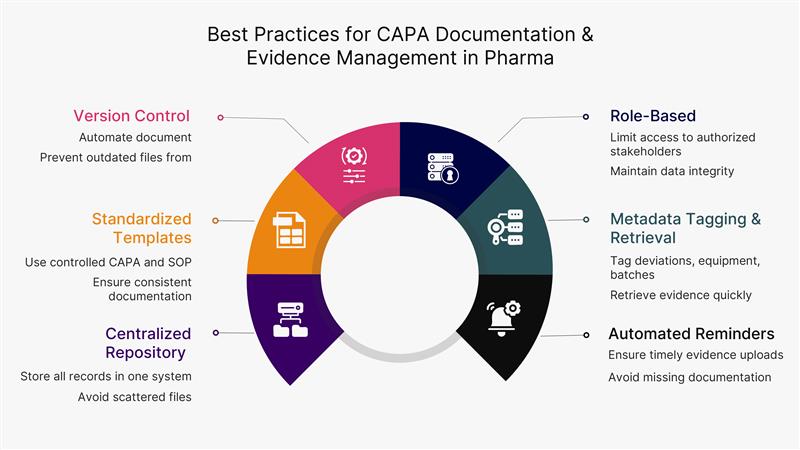
- Standardize all CAPA documents using controlled templates, so investigators no longer have to create data from scratch every time they work.
- Store batch records, logs, and test evidence in a single, structured repository to avoid confusion over file versions and related challenges.
- Use automated document versioning to prevent outdated SOPs or drafts from entering the investigations process.
- Assign role-based access to stakeholders so only qualified stakeholders can upload, modify, or approve CAPA evidence.
- Enable metadata tagging for deviations, equipment, materials, and batches to retrieve evidence instantly during RCA.
- Integrate automated reminders for evidence uploads to ensure everyone stays in the loop and that supporting documents are available during investigations.
3. Cross-functional misalignment and communication gaps
In any industry, CAPA is never executed in isolation; it involves stakeholders from various departments.
Like, in pharmaceuticals, if content uniformity fails for one batch during a routine process check. It triggers deviation and initiates CAPA. Now, solving this would be a combined effort of QA, Production, Engineering, and Supply Chain, and this is where cross-functional collaboration becomes even more important.
Cross-functional gaps not only mislead investigations due to partial or incorrect data but also impact the further course of action (CAPA plan). This results in closure delays of investigation, missed timelines, escalation, or repeated deviations.
Another drawback of siloed processes is that the learning remains isolated in specific teams or departments. And without shared intelligence, organizations miss patterns, leading to repeated deviations and unaddressed systemic risks.
Here’s how teams can address this effectively:
- Create a single cross-functional CAPA workspace so QA, Production, QC, and Engineering work on the same data and updates.
- Use structured or custom forms that automatically route tasks to the right departments to avoid delays during RCA.
- Centralize all investigation files, batch data, equipment records, and more, so every stakeholder views the same evidence and attachments.
- Enable real-time status visibility through shared dashboards so teams can track pending actions, priorities, and deadlines.
- Maintain a shared knowledge repository, so lessons learned, recurring patterns, and systemic risks are accessible across teams.
4. Reactive approach instead of proactive prevention
Many pharmaceutical teams still operate in a reactive mode. They work in a pattern where they only act after an incident or deviation occurs. You may find this a routine task, but this is what amounts to recurring issues.
Simply put, if your CAPA process always triggers after a quality event has already happened, you have already missed many early hints that point to various systemic issues in the quality ecosystem.
Particularly in pharma, the system starts giving warnings much earlier. For example, a slight change in mixing speed, pH value, and temperature; a small system alert on production equipment; or even a steady rise in microbial counts or particle counts in cleanrooms works as an early sign of an upcoming quality event.
A proactive culture continuously scans for these visible signs and encourages teams to investigate them before they become a major issue.
Here’s how to improve this:
- Track recurring incidents using centralized logs, so teams can see emerging trends before they escalate.
- Conduct trend analysis of historical CAPA records to identify these indicators early.
- Use standardized investigation templates, so minor abnormalities are examined with the same focus as significant events.
- Enable cross-module visibility so Production, QA, QC, and Engineering can review risks simultaneously.
- Maintain real-time access to controlled SOPs, so teams never follow outdated instructions that trigger preventable errors.
- Use a structured checklist for routine monitoring to capture weak signals before they become deviations.

Your CAPA Improvement Toolkit Is Ready
Practical resources for faster, better investigation
Download Now5. Lack of digital integration between QMS systems
An integrated system is necessary to centralize the processes going on in silos within the organization.
Without a digital QMS, processes feel disconnected, and evidence and records become scattered across departments. This slows investigations and prevents teams from having a holistic view of impact assessments and risk evaluations.
Let’s assume an operator records a quality event in a standalone deviation form. Without a digital system, the alert logs are separated with no historical linkage, and the reviewer has no visibility into past related deviations unless they manually search for any physical records.
The results?
The investigation starts with an incomplete context; patterns of repeated deviations go missing, and, most importantly, the RCA remains superficial.
While all this is happening in isolation, CAPA actions do not sync with other processes, such as equipment data, training records, etc., which impacts their effectiveness and overall compliance.
Here’s how to fix it:
- Implement a digital, secure quality management system that centralizes and automates your processes, systems, and documents.
- Integrate relevant metadata with quality records, so reviewers can instantly find key details of deviations like batch, equipment, material lot, or stakeholders involved.
- Configure a dashboard that highlights comparative insights about different quality incidents, so investigations start with a full historical context.
- Maintain a unified document library where training files, equipment logs, qualification reports, and SOPs stay connected to their corresponding CAPA actions.
- You can also create real-time visibility dashboards, so QA, Production, QC, and Engineering can see linked records, overdue actions, and risk levels.
- Route CAPA tasks through cross-department workflows that notify stakeholders automatically, preventing delays caused by manual follow-ups.
Elevating Pharma Quality Through Better Systems
In the pharmaceutical industry, a failed CAPA not only exposes the company to compliance risks but also impacts the quality of the drug, patient safety, global drug supply, and even operational consistency.
However, the path forward is clear: pharmaceutical companies have to make a significant shift in their approach—from reactive, problem-solving to proactive, data-driven. And going digital with a centralized, secure QMS can be a transformative step in this direction.
BizPortals QCFlow, a robust quality management system, helps eliminate the very challenges that keep CAPA systems weak and inconsistent. With effective document control features such as document versioning, metadata integration, audit trails, e-signature, and permission-based access, it ensures that all your CAPA records and SOPs are up to date, accessible, and traceable.
While it offers cross-module integration to ensure seamless data flow and visibility across departments, it also enables role management to prevent unauthorized access. BizPortals QCFlow, a fully configurable solution, empowers users to create different templates, forms, fields, and even an entire custom module to fit your specific requirements.
Want a closer look at BizPortals QCFlow? Join a live demo session to go through the features, talk about your requirements, and get a clear picture of how everything works.




